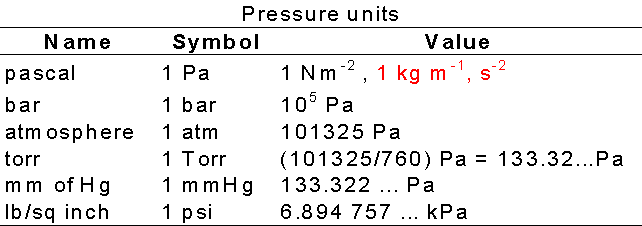
Atmospheres and Conversions: 1.00 atm = x 10 5 Pa = kPa = 760. Torr = 14.7 psi To convert pressure: 1.Turn what you have to atm 2.Multiply. - ppt download

GASES. Identify the abundances of the naturally occurring gases in the atmosphere. Describe the historical development of the measurement of pressure. - ppt download







/sun-beams-diffuse-ocean-micronesia-palau-128941208-587b91ad5f9b584db36312c9.jpg)











![Solved]: 1. The common units for the units of pre Solved]: 1. The common units for the units of pre](https://media.cheggcdn.com/study/698/698d56ec-48f4-4912-b705-f36df5b08db9/image)