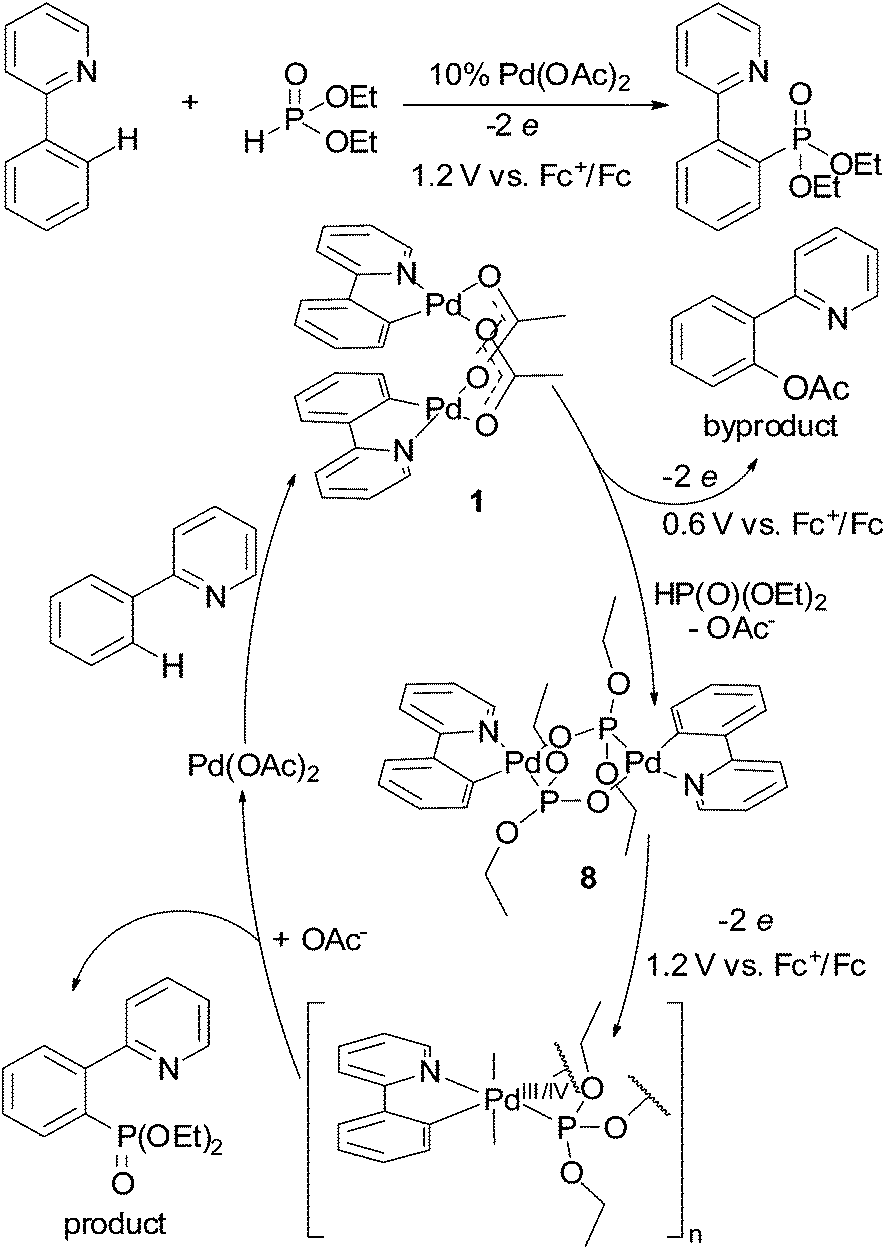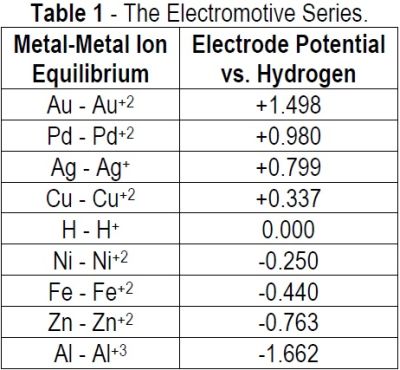
Redox trends in cyclometalated palladium( ii ) complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT03786K
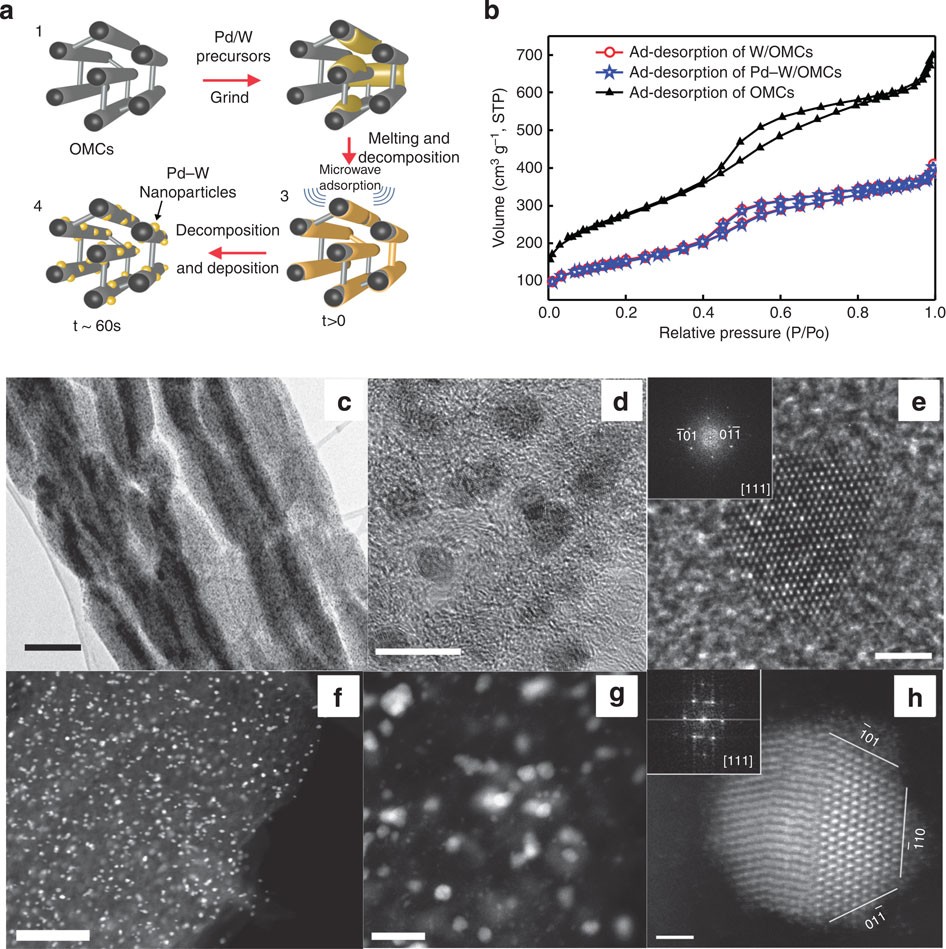
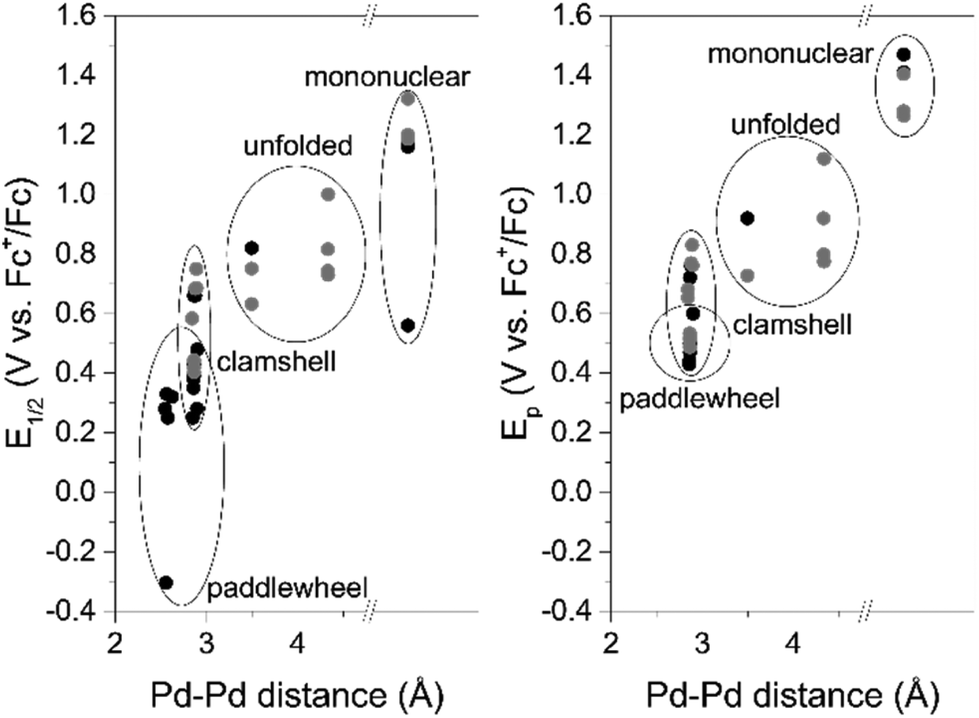
Small palladium islands embedded in palladium–tungsten bimetallic nanoparticles form catalytic hotspots for oxygen reduction | Nature Communications

Electrochemical synthesis of palladium (Pd) nanorods: An efficient electrocatalyst for methanol and hydrazine electro-oxidation - ScienceDirect
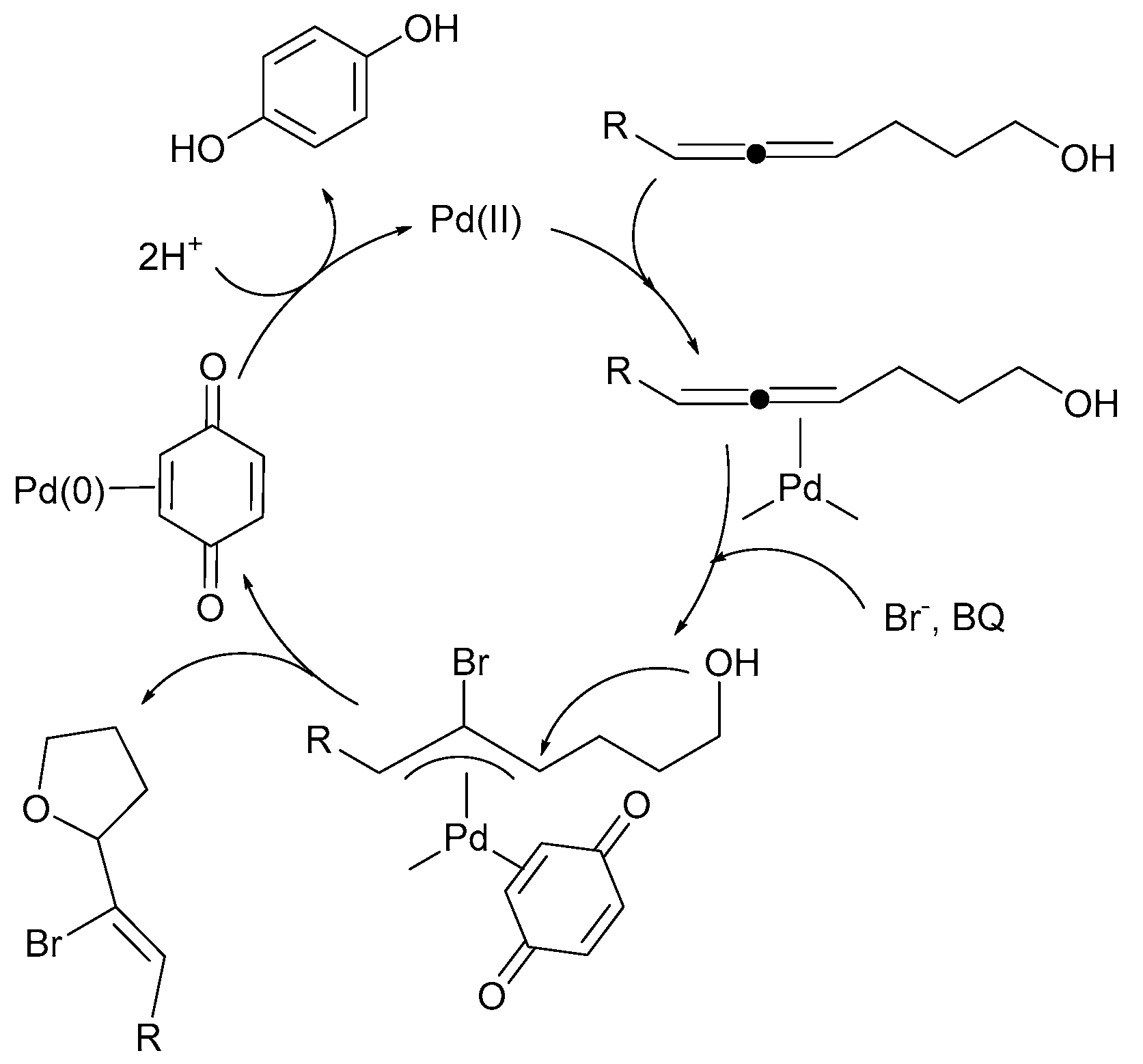
Inorganics | Free Full-Text | Redox-Induced Aromatic C–H Bond Functionalization in Metal Complex Catalysis from the Electrochemical Point of View | HTML

Enhanced Electrocatalytic Reduction of Oxygen at Electrodes Coated with a Multi-Metallic Co(II)/Pt(II) Porphyrin

1 21 Electrochemistry. 2 Counting Electrons: Coulometry and Faraday's Law of Electrolysis Example 21-1: Calculate the mass of palladium produced by the. - ppt download

Palladium Oxidase Catalysis: Selective Oxidation of Organic Chemicals by Direct Dioxygen‐Coupled Turnover - Stahl - 2004 - Angewandte Chemie International Edition - Wiley Online Library

Electrochemical behavior of CO2 reduction on palladium nanoparticles: Dependence of adsorbed CO on electrode potential - ScienceDirect

Cyclic voltammetry during FA electrooxidation on palladium (a) with... | Download Scientific Diagram

Table 1 from Nanoscale zero valent iron and bimetallic particles for contaminated site remediation | Semantic Scholar

Merging Electrolysis and Nickel Catalysis in Redox Neutral Cross-Coupling Reactions: Experiment and Computation for Electrochemically Induced C–P and C–Se Bonds Formation | CCS Chem
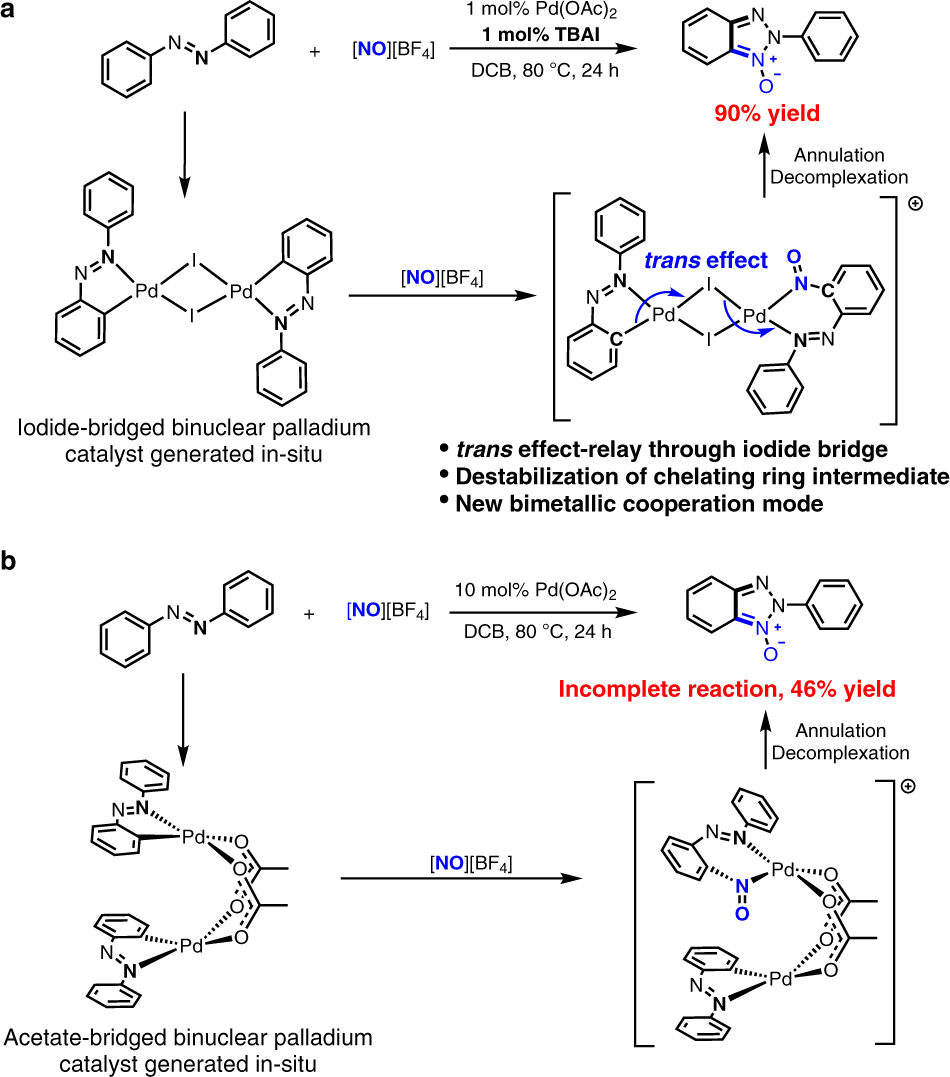
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry

Electrochemical pH sensor based on a hydrogen-storage palladium electrode with Teflon covering to increase stability - ScienceDirect

Standard reduction potential of possible cathode reaction and redox... | Download Scientific Diagram